+86-18343147735
+86-18343147735




In Vitro Diagnostic (IVD) products are essential in modern healthcare. They play a critical role in disease identification and management. For global buyers, navigating the complexities of the IVD market can be daunting. This landscape is filled with various products, suppliers, and regulations. It is vital to make informed decisions to ensure quality and reliability.
Understanding the nuances of IVD systems is key. Buyers should be aware of certification standards and product sourcing. This awareness can prevent costly mistakes. Many IVD products claim to offer accuracy and efficiency. However, not all meet these expectations. Engaging with trusted manufacturers and understanding their expertise is crucial in this process.
Global buyers must also consider market trends and innovations. New technologies emerge rapidly in the IVD space. Staying informed can enhance purchasing strategies. Reflection on past purchasing experiences can guide future decisions. Being knowledgeable in the IVD domain not only boosts confidence but also ensures better patient outcomes.


In vitro diagnostics (IVD) play a crucial role in modern healthcare. They enable rapid and accurate disease detection. This is especially important in areas with limited access to healthcare facilities. By analyzing samples outside the body, healthcare providers can make informed decisions quickly. Timely diagnosis can significantly alter treatment outcomes.
Understanding the nuances of IVD is essential. The reliability of test results can vary widely. Factors such as sample quality and testing methods influence these results. Buyers must be cautious when selecting diagnostic tools. Missteps in choosing the right equipment can lead to misdiagnoses. This can have serious implications for patient care.
Moreover, continuous education is vital. Staying updated on innovations in IVD is necessary. It ensures that products meet changing healthcare demands. Engaging with healthcare professionals can provide valuable insights and foster trust. In a rapidly evolving field, there is always room for improvement and reflection on practices. Embracing a culture of learning can enhance the effectiveness of IVD in global healthcare.
When selecting in vitro diagnostic (IVD) products, the importance of quality cannot be overstated. Buyers must assess the accuracy and reliability of the tests. Look for certifications that indicate rigorous testing and compliance with standards. This helps ensure the products perform as expected in clinical settings.
Cost is another key factor. While it can be tempting to choose the lowest-priced option, this may not always guarantee quality. Analyze the long-term cost-effectiveness of the products. Sometimes, investing a little more upfront can lead to better results and fewer repeat tests in the future. Balancing cost and quality is essential.
Lastly, consider the supplier's support and service. Reliable support offers peace of mind. A good supplier provides training, troubleshooting, and timely responses to inquiries. This can make a significant difference, especially when urgent issues arise. Buyers might encounter delays or miscommunications. Assessing the level of support available can alleviate potential complications.
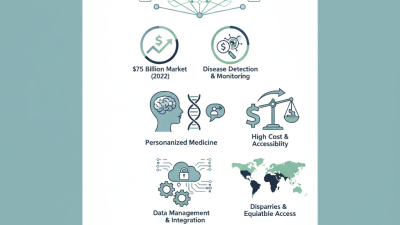
When evaluating in vitro diagnostics (IVDs), regulatory compliance is paramount. Globally recognized standards, such as ISO 13485, guide the quality management systems for medical devices. Compliance with these standards helps ensure that products are safe and effective. A report by MarketsandMarkets indicates that the global IVD market is projected to reach $105.9 billion by 2025. This growth underscores the importance of adhering strictly to regulatory requirements.
Quality standards are not only about meeting initial benchmarks. Continuous monitoring is essential. Manufacturers must regularly validate their processes and products. For instance, the FDA mandates annual reports for many IVDs to maintain transparency and efficiency. However, not all companies prioritize this ongoing compliance. A survey found that nearly 30% of buyers were unsure about the regulatory status of their suppliers. This gap can lead to significant risks, including product recalls and financial losses.
It's critical for buyers to ask about compliance documentation and quality control measures. They should investigate certifications, audits, and past performance. A focus on these aspects can mitigate risks. It is essential to realize that achieving compliance is an ongoing journey, not just a checklist. Companies must be prepared to adapt to changing regulations and emerging technologies.
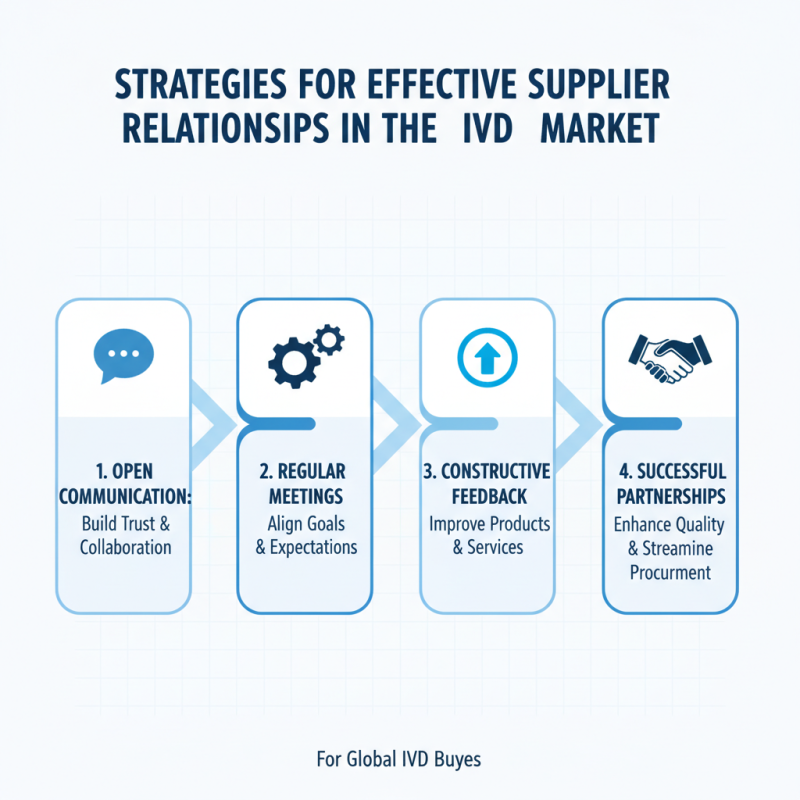
Building effective supplier relationships in the in vitro diagnostic (IVD) market is crucial for global buyers. Successful partnerships can enhance product quality and streamline procurement processes. Open communication serves as the foundation for trust and collaboration. Regularly scheduled meetings can ensure alignment on goals and expectations. Suppliers appreciate feedback, so sharing insights can help improve products and services.
One practical tip is to perform thorough supplier assessments. Evaluate their capabilities and reliability. This helps in identifying potential risks before they affect your operations. Establishing clear contractual agreements is essential. This sets the groundwork for accountability and performance standards. Frequent reviews of these agreements can help adjust to changing market dynamics.
Flexibility plays a key role in supplier relationships. Market conditions can shift rapidly. Being prepared to adapt can benefit both parties. However, not every partnership will be perfect. Learn from challenges and setbacks. Reflecting on issues can pave the way for more effective solutions. Incorporating continuous improvement practices will strengthen future collaborations. Fostering a willingness to share both successes and failures builds resilience in partnerships.
In the dynamic field of in vitro diagnostics (IVD), staying ahead requires understanding emerging market trends. The rapid advancement in technology has shifted the focus toward automation, artificial intelligence, and point-of-care testing. These innovations drastically improve efficiency and accuracy, which is crucial for medical professionals. Buyers must adapt to these shifts to enhance patient outcomes.
Collaboration is also vital in navigating IVD advancements. Engaging with research institutions can provide valuable insights into cutting-edge technologies. However, not every innovation will suit all markets. Understanding regional healthcare needs is crucial. Local regulations and cultural factors often dictate what solutions are viable. Ignoring these aspects can lead to missteps in product selection.
Market research is essential for good decision-making. Analyzing competitor offerings can reveal gaps in the market. It’s important to remain adaptable and open to feedback. While new technologies may seem promising, they require careful evaluation. Prioritizing investments in solutions that meet real-world needs will lead to better healthcare outcomes and sustainable growth.