+86-18343147735
+86-18343147735




In recent years, glucosamine has gained significant attention for its potential health benefits, especially in joint support. The demand for glucosamine products has surged in China, leading to a variety of quality assessments being conducted. The Glucosamine Test plays a crucial role in ensuring that consumers receive effective and safe supplements.
This article explores the top 10 glucosamine tests currently utilized in China. Each test varies in methodology and accuracy, highlighting the need for reliable assessments. Many companies strive to enhance quality, yet some challenges remain. Not all testing methods provide the same level of assurance, which can confuse consumers.
In an ever-growing market, understanding these tests is vital. Consumers deserve confidence in what they purchase. Awareness of testing standards helps mitigate risks. Evaluating glucosamine's efficacy is not just about marketing. It is about consumer health and safety.

Glucosamine is a natural compound found in cartilage. It plays a crucial role in joint health and pain relief. Many people use glucosamine supplements to manage arthritis or joint-related discomfort. The effectiveness of these supplements largely depends on their quality. Ensuring that glucosamine products meet high standards is vital for consumer trust.
In China, quality assessment is paramount. Various tests are conducted to evaluate purity and potency. These assessments look for contaminants and measure active ingredient concentrations. When products fail to meet quality benchmarks, consumers may question their efficacy. This creates a gap between expectation and reality. Without strict oversight, substandard products may emerge in the market.
The importance of glucosamine in joint health cannot be overlooked. However, quality assessment methods must improve continuously. There are inconsistencies in testing protocols across regions. This inconsistency can lead to confusion among consumers. A reliable glucosamine test should be comprehensive, covering various aspects of quality. By addressing these challenges, stakeholders can enhance product reliability and consumer satisfaction.
Glucosamine is widely used for joint health. However, testing methods for its quality in China vary significantly. Industry reports indicate that the market for glucosamine is projected to reach 1.4 billion USD by 2025. With the increasing demand, effective testing methods are essential for ensuring product purity and efficacy.
High-performance liquid chromatography (HPLC) is a common method for glucosamine testing. It allows for precise measurement of glucosamine content. This technique is critical for detecting impurities. Near-infrared spectroscopy (NIRS) is another method gaining traction. It offers rapid analysis and does not require complex sample preparation. Such advancements are vital in a market that lacks uniform standards.
Despite the growth of testing technologies, challenges remain. Many laboratories still lack the latest equipment. Standards for glucosamine testing are not universally adopted. The consistency of results can vary between institutions. Ongoing training for laboratory staff is necessary to enhance accuracy. Furthermore, public awareness about testing quality is crucial. Consumers deserve reliable information for their health choices. This topic warrants further discussion and scrutiny in the industry.
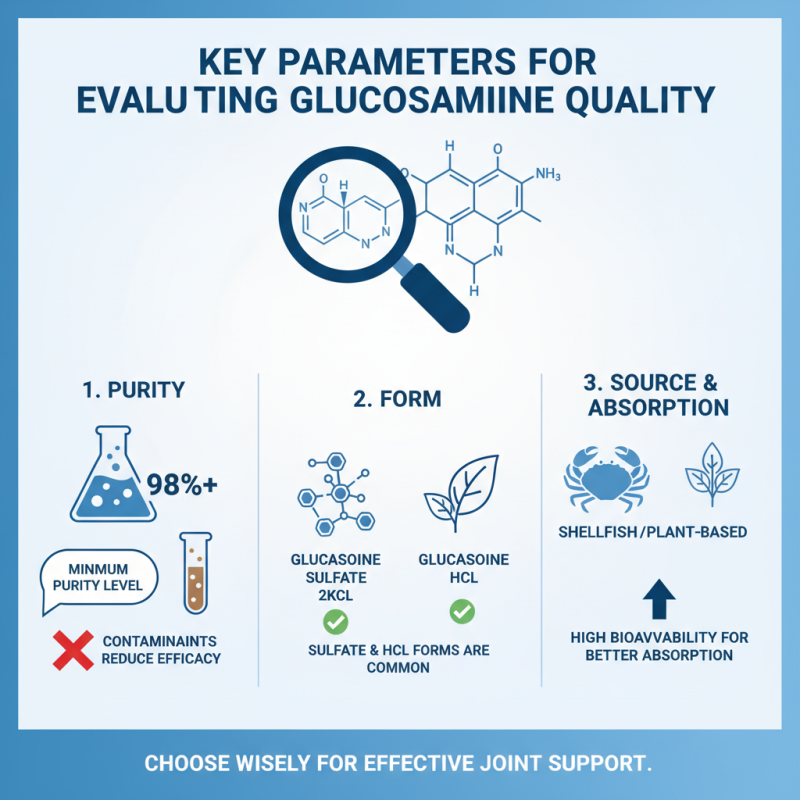
When assessing the quality of glucosamine, several key parameters come into play. Firstly, purity is critical. High-quality glucosamine should have a purity level of at least 98%. Lower purity levels often indicate contamination or adulteration. Impurities can lead to health risks and diminished efficacy.
Another essential factor is the source of glucosamine. It can be derived from shellfish, or it can be synthesized. Shellfish-derived glucosamine may cause allergic reactions in some individuals. On the other hand, synthetic versions provide a consistent and hypoallergenic option. Organizations like the World Health Organization emphasize the importance of thorough documentation regarding the source and manufacturing process.
Tips: Always check for third-party certifications. These can assure that the product meets industry standards and safety guidelines. Look for lab analysis results that confirm both purity and biological activity.
Beyond purity and source, bioavailability is a pivotal indicator of glucosamine quality. It refers to the degree and rate at which an active ingredient or active moiety is absorbed. Enhanced bioavailability means more effective results.
Finally, transparency is vital. Manufacturers should provide clear information about ingredient testing and quality assessments. However, some companies may lack sufficient transparency, making it hard to discern quality. Always prioritize products that offer comprehensive testing data. This vigilance ensures you select a safe and effective glucosamine supplement.
In China, glucosamine testing is crucial for quality assessment in supplements. Various tests are commonly used to ensure purity and efficacy. One prominent method is High-Performance Liquid Chromatography (HPLC). This test can effectively separate and identify glucosamine in complex mixtures. It provides detailed insights into the compound's concentration.
Another key test is Infrared Spectroscopy (IR). IR helps in determining the quality of glucosamine by analyzing its molecular structure. This method is quick and provides immediate results. Laboratories often utilize this technique alongside HPLC for comprehensive analysis.
Tips: Always verify the calibration standards used in equipment. Regular maintenance ensures accurate readings. Additionally, consider running control samples to validate results. Researchers should also regularly update their methodologies based on the latest scientific findings.
Despite advancements, challenges remain. Variability in raw material sources can affect results. This requires stringent testing protocols. Laboratories need to establish robust quality control measures. These improvements will enhance the reliability of glucosamine testing in China. Taking these actions will ultimately benefit manufacturers and consumers alike.
In China, the evaluation of glucosamine products is guided by specific regulatory standards. These standards focus on the chemical composition, purity, and safety of glucosamine. Laboratories conduct tests to determine the presence of contaminants and verify the concentration of active ingredients. Proper methodologies must be employed to ensure accuracy and reliability.
Testing involves various techniques, such as high-performance liquid chromatography (HPLC) and mass spectrometry. These methods provide detailed insights into the product's quality. However, challenges remain. Many labs still lack adequate training and equipment. This raises concerns about data integrity in testing outcomes. Some manufacturers may prioritize profit over compliance.
Additionally, consumers often lack awareness of these testing protocols. They may unknowingly purchase substandard products. Continuous education on quality assessment is essential. Transparency in testing results must also improve. Addressing these gaps will foster a more robust glucosamine market in China.
| Test Name | Purpose | Standard Method | Acceptance Criteria | Frequency of Testing |
|---|---|---|---|---|
| HPLC Analysis | Purity Assessment | GB/T 19903-2019 | ≥ 95% | Every Batch |
| GC-MS Analysis | Contaminant Detection | ISO 17025 | Not detectable | Every Batch |
| Microbial Testing | Safety Assessment | GB 4789.2 | Absent | Every Batch |
| Heavy Metal Analysis | Toxic Element Evaluation | GB 5009.74 | < 10 ppm | Every Batch |
| Water Content Analysis | Stability Assessment | GB/T 6009 | ≤ 5% | Every Batch |
| pH Testing | Formulation Stability | GB/T 6368 | 6.0 - 7.5 | Every Batch |
| Ash Content Analysis | Content Impurity | GB/T 5009.4 | ≤ 2% | Every Batch |
| Stability Testing | Shelf Life Evaluation | ISO 22000 | Stable for 2 years | Every Year |
| Viscosity Testing | Quality Consistency | GB/T 5561 | Consistent Results | Every Batch |